Printed circuit boards (PCBs) serve as fundamental components of electronic products, playing a crucial role in contemporary technology.
With the advancement of 5G technology, communication systems demand ever-increasing speeds and capacities.
This demand imposes stricter specifications on PCB substrate characteristics, including thickness, surface morphology, and impedance.
During PCB manufacturing, electroplated copper forms conductive pathways and interconnect structures, with coating quality directly determining the board’s electrical performance and reliability.
Acidic Copper Sulfate Plating and the Function of Additives
The acidic copper sulfate plating solution system remains the mainstream approach for PCB copper plating.
This system employs composite additives to regulate copper deposition behavior, ensuring uniform electroplated copper layers on PCB electrical interconnect surfaces.
Generally, adding only a small amount of additives to the plating solution can significantly enhance both solution and deposit performance.
This approach achieves uniform plating within through-holes and overfilling of blind vias.
Investigating the mechanisms of copper plating additives and developing high-performance additives represents one of the pressing challenges in the PCB industry.
Research Focus on DPS Acceleration Mechanisms
Researchers have extensively studied sodium N, N-dimethyl-dithiocarbamoylpropane sulfonate (DPS) as an accelerator for acidic copper sulfate electroplating.
However, they still do not completely understand the adsorption mechanism of DPS on copper surfaces or how it accelerates copper electrodeposition.
This study systematically investigates the adsorption behavior of DPS molecules on copper surfaces through a combination of electrochemical testing and theoretical calculations.
The study examines the influence of DPS molecular end-group structures on its performance.
It aims to deepen the understanding of the accelerator’s molecular-level mechanism of action and to provide a theoretical basis for developing novel accelerators.
Experiment
Plating Solution Formulation
The base plating solution (VMS) comprised 220 g/L CuSO₄·5H₂O, 50 g/L sulfuric acid (98% H₂SO₄), and 60 mg/L Cl⁻.
The inhibitor was 300 mg/L polyethylene glycol (PEG, molecular weight 8,000), with DPS added as an accelerator.
To minimize interactions between additives and investigate the mechanism of DPS more clearly, we did not add a leveling agent to the formulation in this study.
Electrochemical Testing
We conducted electrochemical testing at room temperature using the AUT84480 electrochemical workstation manufactured by Metrohm China Co., Ltd.We employed a three-electrode system.
It featured a 3.0 mm diameter Pine rotating disk platinum electrode (Pt-RDE) as the working electrode, a platinum sheet as the counter electrode, and a saturated mercury/mercury sulfate electrode (SMSE) as the reference electrode.
Before experimentation, the working electrode underwent polishing and ultrasonic cleaning.
Subsequently, it was subjected to 20 cycles of cyclic voltammetry (CV) in a 1.0 mol/L sulfuric acid solution using the three-electrode system.
The potential range was −0.4 to 0.6 V (vs. SMSE), with a scan rate of 50 mV/s, ensuring a smooth surface and absence of copper dissolution peaks.
We conducted both cyclic voltammetry (CV) and linear sweep voltammetry (LSV) at a scan rate of 50 mV/s.
We determined the current density applied during constant-current tests based on the specific system.
Before performing constant-current tests, we deposited copper onto the Pt-RDE surface using VMS at room temperature for 600 s to prepare the Cu-RDE working electrode.
Theoretical Calculations
1. Quantum Chemical Calculations for DPS
We visualized the DPS molecular structure using Gauss View software and optimized it with Gaussian 09.
Based on the optimized configuration, we visualized the molecular orbitals using IQmol and the molecular surface electrostatic potentials using VMD software.
2. Molecular Dynamics Simulation of DPS
We conducted molecular dynamics simulations using Materials Studio software.
We selected a structurally stable Cu(111) crystal plane as the copper surface model.
The optimized DPS molecules were mixed with water molecules at a 1:500 molar ratio and adsorbed onto the copper surface to form an adsorption box.
We simulated NVT ensemble conditions with a cutoff radius of 9.5 nm and a time step of 1 fs, for a total of 500 ps.
Results and Discussion
Effect of DPS on the Electrochemical Behavior of Copper Electrodeposition
1. Cyclic Voltammetry Analysis
To investigate the effect of DPS concentration on copper electrodeposition behavior, we conducted CV tests on VMS + 300 mg/L PEG plating solutions containing different DPS concentrations.
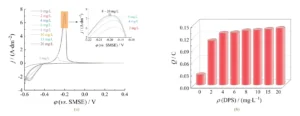
As shown in Figure 1a, reduction peaks and oxidation peaks appeared during negative and positive scans, respectively, corresponding to the reduction of Cu²⁺ to Cu⁰ and the anodic stripping of Cu.
During the forward scan, the copper stripping peak emerges near the self-potential of −0.41 V, indicating the onset of copper dissolution.
This peak vanishes when the copper on the electrode surface is completely dissolved.
The stripping charge of the copper peak, defined as Q, characterizes the deposition amount of copper during electroplating .
Generally, a larger copper stripping peak indicates faster copper deposition.
To analyze the effect of DPS concentration on the copper deposition rate more intuitively, we integrated the stripping peaks for Cu on each curve in Figure 1a, producing Figure 1b.
This shows that as the DPS mass concentration increases from 0 mg/L to 6 mg/L, Q rapidly rises from an initial 0.025 0 C to 0.129 3 C;
As DPS mass concentration further increases, the change in Q flattens, indicating that the acceleration effect of DPS stabilizes.
This occurs because at lower DPS concentrations, DPS rapidly occupies active sites that accelerate deposition, significantly boosting copper deposition rates.
When the DPS mass concentration exceeded 6 mg/L, the active sites reached saturation.
At this point, the copper deposition rate was primarily limited by the number of active sites rather than the DPS concentration.
Concurrently, as the DPS concentration increased, the copper deposition potential shifted positively, further reflecting the enhanced deposition rate.
2. Linear Sweep Voltammetry Analysis
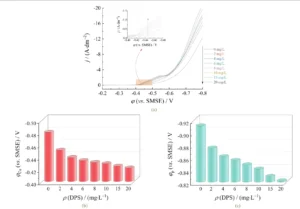
Figure 2 shows the LSV curves of copper electrodeposition with different DPS concentrations added to the VMS + 300 mg/L PEG plating solution.
It also shows the corresponding copper electrodeposition onset potential (Cu) and the reduction peak potential (p), which refers to the potential at the maximum reduction peak current density of Cu²⁺ reduction to Cu⁰.
Compared to the system without DPS, the addition of 2 mg/L DPS resulted in a significant positive shift in the copper deposition onset potential, indicating that DPS promotes copper deposition.
As shown in Figure 2b, increasing the DPS mass concentration from 0 mg/L to 20 mg/L progressively shifted the copper electrodeposition onset potential from −0.48 V to −0.42 V.
This indicates that within this concentration range, the acceleration effect of DPS intensifies with increasing concentration.
Figures 2a and 2c show that the reduction peak potential of copper electrodeposition shifts positively as the DPS mass concentration increases.
This observation further confirms DPS’s significant acceleration effect on copper deposition.
Overall, DPS effectively promotes copper electrodeposition, and its acceleration effect intensifies within the 0–20 mg/L mass concentration range, consistent with the CV analysis results.
3. Chronopotentiostatic Analysis
In constant-current tests, changes in polarization potential can be used to evaluate the effect of additives on copper electrodeposition.
A more negative polarization potential indicates stronger polarization, signifying greater resistance to copper deposition.
To investigate the effect of additives on copper electrodeposition, VMS was employed during the initial phase of the constant-potential test.
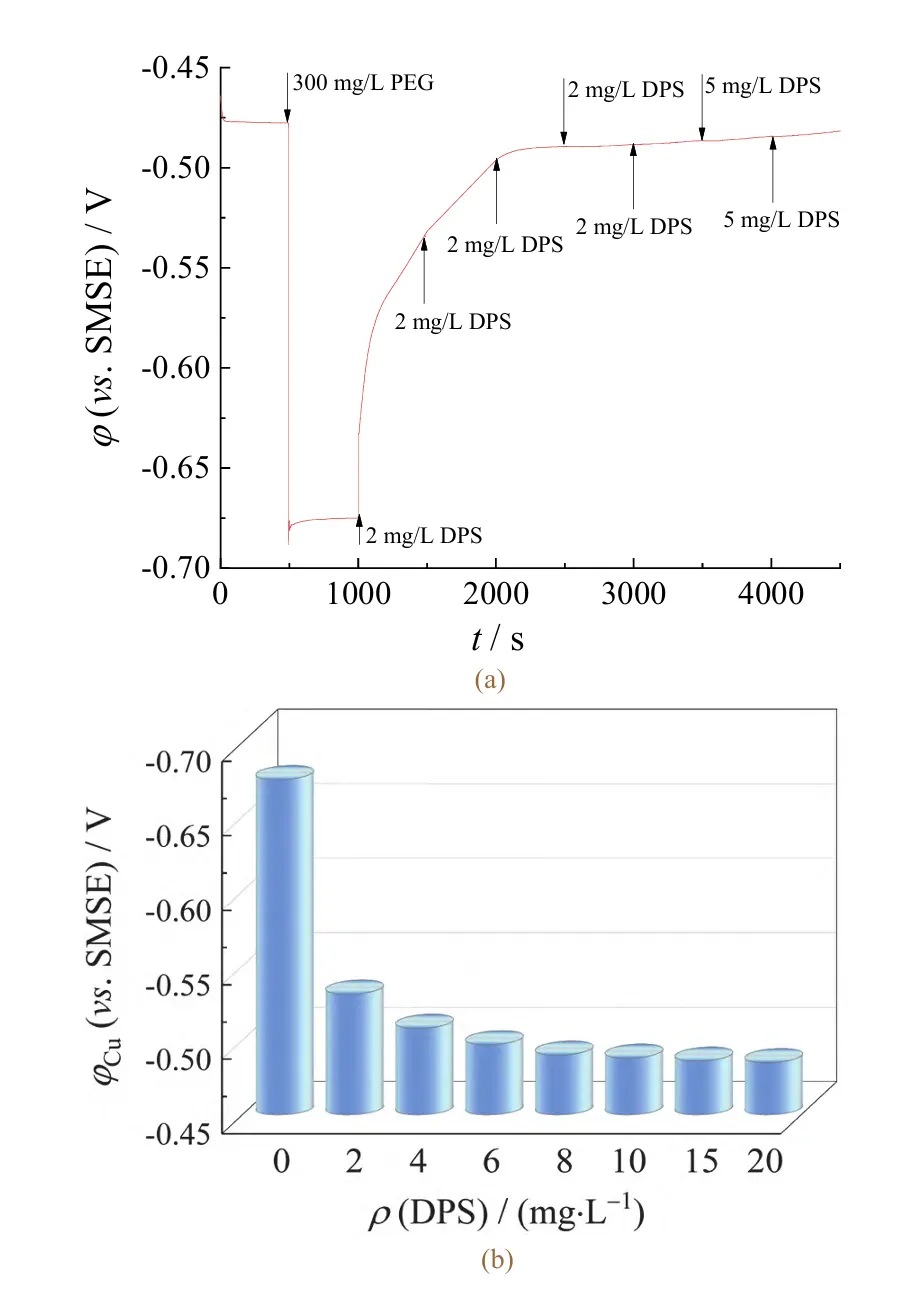
At 500 s into the test, 300 mg/L PEG was added. Starting at 1000 s, DPS was added at specific intervals of 500 s, achieving a total mass concentration of 20 mg/L DPS by 4000 s.
As shown in the constant potential test results in Figure 3a, the copper electrodeposition potential in VMS was −0.477 V.
After adding 300 mg/L PEG, it shifted negatively to −0.675 V, indicating enhanced cathode polarization.
The most direct cause was the formation of an adsorption layer on the copper surface by PEG interacting with Cu²⁺ and Cl⁻ ions.
This adsorption layer impedes copper ion diffusion, inhibiting the copper electrodeposition process.
Upon further addition of 2 mg/L DPS to the electrolyte, the potential shifted significantly positive to −0.531 V (as shown in Figure 3b), indicating markedly weakened cathode polarization.
DPS adsorbs onto the copper surface at one end, while its free sulfonic acid group (─SO₃H) remains in solution to capture Cu²⁺ ions.
This process accelerates Cu²⁺ transport, thereby reducing cathode polarization and promoting copper deposition.
As DPS concentration increases, the potential continues to shift positively, indicating strong antagonism between DPS and PEG.
This may occur because higher DPS concentrations increase the number of DPS molecules in solution.
These molecules compete with PEG molecules, gradually displacing PEG from its adsorption sites on the copper surface, thereby weakening PEG’s inhibitory effect on copper electrodeposition.
Theoretical Calculation and Analysis of DPS Influence on Copper Electrodeposition
1. Quantum Chemical Analysis of DPS
To elucidate the DPS acceleration effect and the influence of its concentration observed in the electrochemical tests, this study uses a combined approach of quantum chemical calculations and molecular dynamics simulations.
These methods allow analysis at the molecular scale.
According to frontier molecular orbital theory, the adsorption capacity of organic molecules on metal surfaces is closely related to their energy of the highest occupied molecular orbital (EHOMO), energy of the lowest unoccupied molecular orbital (ELUMO), and energy gap (∆E = ELUMO − EHOMO).
Generally, a lower EHOMO and higher ELUMO indicate stronger bonding between organic molecules and metal surfaces.
A smaller ∆E value signifies enhanced adsorption capacity of organic molecules onto metal surfaces.
Quantum chemical calculations provide electronic structure parameters and frontier molecular orbital information for accelerator molecules.
These calculations enable the prediction of adsorption sites and help elucidate the relationship between molecular structure and adsorption mechanisms.
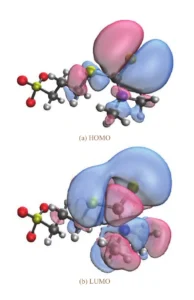
Figure 4 displays the HOMO and LUMO distributions of DPS obtained from quantum chemical calculations, where yellow, red, blue, black, and white spheres represent S, O, N, C, and H atoms, respectively.
The calculated EHOMO of DPS is −0.047 96 eV, the ELUMO is −0.223 85 eV, and the energy gap ΔE is 0.175 89 eV.
As shown in Figure 4, the HOMO and LUMO of DPS are primarily distributed on the N atom of the dimethylamino group and the adjacent S atom, indicating that the N atom is the main adsorption site of DPS on the copper surface.
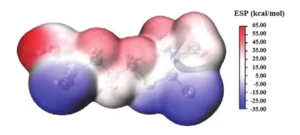
2. Electrostatic Potential Mapping of DPS
Electrostatic potential (ESP) can be used to predict molecular properties and visualize reaction sites.
In ESP maps, regions of high electron density are typically rendered red, indicating nucleophilic reactions; regions of low electron density appear blue, indicating electrophilic reactions.
Figure 5 shows the ESP distribution of DPS.
The red and pink regions mainly surround the nitrogen (N) atom of the dimethylamino group, while the blue regions are concentrated around the sulfur atom of the sulfonic acid group and the C=S bond.
This distribution is closely related to the lone pair of electrons present on the sulfur atom.
These results indicate that the dimethylamino group may act as the primary adsorption site for DPS on the copper surface.
3. Electrostatic Potential and Adsorption Sites
Meanwhile, the sulfonic acid group and C=S bond can more readily bind with copper ions, thereby accelerating copper deposition.
This provides an electronic structural explanation for why DPS strongly adsorbs to the copper surface via this terminal group.
It also explains why DPS competes with PEG for adsorption sites, consistent with the constant-current test results.
4. Molecular Dynamics Simulations of DPS Adsorption
To gain deeper insight into the adsorption process of DPS on the copper surface, molecular dynamics simulations were further conducted.
Figure 6 illustrates the adsorption states of DPS molecules at 0 ps, 5 ps, and 100 ps within the established box. Yellow, red, blue, black, and white spheres represent S, O, N, C, and H atoms, respectively.
As shown in Figure 6a, at 0 ps, the DPS molecule is located at the center of the box.
Figure 6b shows that at 5 ps, the DPS molecule begins to contact the Cu(111) surface, with the dimethylamino group being the first to adsorb onto the copper surface.
Figure 6c reveals that at 100 ps, the DPS molecule adsorbs onto the Cu(111) surface in an “upright” configuration.
At this stage, the dimethylamino end anchors to the copper surface while the sulfonate group faces the solution.
This orientation enables the sulfonate group to capture Cu²⁺ ions from the solution more efficiently, thereby accelerating the copper electrodeposition process.
This result provides a kinetic explanation for the reduced copper deposition overpotential observed in the LSV curve of Figure 2.
This finding is consistent with the conclusions from the electrostatic potential analysis.
Together, they confirm the microscopic mechanism by which DPS promotes copper electrodeposition through its specific adsorption configuration.

Conclusion
DPS demonstrates outstanding acceleration performance in copper electrodeposition due to its unique molecular structure and adsorption mechanism.
It offers a novel solution for copper plating processes in PCB manufacturing.
Integrated electrochemical testing and theoretical calculations show that DPS strongly adsorbs onto copper surfaces via its terminal dimethylamino groups, competing with the inhibitor PEG for binding sites.
Simultaneously, its sulfonic acid groups at the opposite end face the solution, effectively capturing Cu²⁺ ions.
This unique adsorption-transport synergistic mechanism is the fundamental reason for its significant acceleration of copper electrodeposition.


