The principle of acid etching relies on the reaction between divalent copper in the etching solution and the metallic copper on the surface of the printed circuit board (PCB).
The reaction produces monovalent copper, which forms a complex with high-concentration chloride ions and remains stable within the etching solution.
The reaction equation is: CuCl₄²– + Cu + 2Cl⁻ = 2CuCl₃²–.
Based on this chemical equation, free Na⁺, K⁺, and NH₄⁺ ions in the etching solution do not participate in the etching reaction.
From a chemical reaction kinetics perspective,
using sodium chloride, potassium chloride, or ammonium chloride at equivalent molar concentrations as the chloride salt in the etching solution does not affect the etching rate.
Experiments reveal that replacing sodium chloride with equivalent amounts of ammonium chloride or potassium chloride significantly increases the etching rate.
The theory of negative correlation between etching rate and viscosity in etching solutions explains this phenomenon well.
Viscosity represents the macroscopic manifestation of interactions between ions and molecules within a solution.
Different solutes interact with water molecules through varying degrees of hydration, hydrolysis, and hydrogen bonding.
Stronger interactions increase ion migration resistance and elevate solution viscosity.
This physical property of etching solution viscosity reflects the solution’s fluidity and diffusion capacity, directly influencing mass transfer processes in the etching reaction.
Lower etching solution viscosity effectively reduces the “pooling effect” during etching, promoting contact between fresh etchant and the copper metal surface.
Additionally, under identical spray pressure conditions in the etching machine, lower viscosity yields higher spray flow rates, indicating that reduced viscosity facilitates increased etching rates.
Therefore, incorporating viscosity parameters and conducting targeted research holds significant importance for refining etching theory.
Experiment
Instruments, Materials, and Chemicals
Electronic analytical balance (JA3003),
Pen viscometer (0.4 mm, constant 0.001 896 mm²/s²),
The experiment requires a hydrometer (with ranges of 1.0–1.1, 1.1–1.2, and 1.2–1.3) and a stopwatch.
It also requires a water bath equipped with an electromagnetic stirrer.
The materials include double-sided copper-clad laminate (50 mm × 50 mm square, copper thickness 70 μm).
They also include copper chloride dihydrate (analytical grade), concentrated hydrochloric acid (analytical grade), sodium chloride (analytical grade), potassium chloride (analytical grade), ammonium chloride (analytical grade), and polyethylene glycol 6000 (analytical grade).
Experimental Design
1. Viscosity Measurement
(1). Concept of Viscosity
Using a Pennington capillary viscometer, the kinematic viscosity υ (mm²/s) of a solution can be precisely measured.
Kinematic viscosity primarily reflects the strength of viscous forces within the solution.
(2) Principle of Viscosity Measurement
Poiseuille’s law describes the principle by which a Poiseuille viscometer measures fluid viscosity.
The expression for the volumetric flow rate Q through the capillary is as follows:
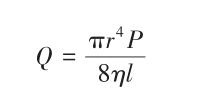
In the equation: r is the capillary radius, m;
P is the pressure difference across the liquid column in the capillary, Pa;
l is the capillary length of the Pycnometer, m. The expression for dynamic viscosity η is as follows:
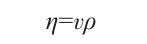
In the equation: ρ is the density of the solution, g/cm³; v is the kinematic viscosity of the solution.
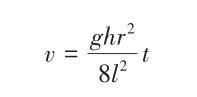
In the equation: g is the gravitational acceleration, m/s²; t is the time taken for the sample to pass through the capillary tube of the Viscometer, s; h is the liquid level height, m.
Here, r, l, and h are fixed values, while t is solely related to υ and is proportional to it.
Therefore, under identical experimental conditions, the duration of passage accurately reflects the magnitude of υ.
Equation (3) defines the constant C of the pycnometer.
For practical application, the mathematical units of the Pycnometer constant are typically converted to mm²/s² (the constant marked on the instrument is calibrated by the manufacturer using standard liquids of known viscosity).
When measuring with a Pycnometer, the sample volume affects the liquid level difference h.
Therefore, during the experiment, an equal volume of the solution sample must be taken for each measurement.
(3) Viscosity Measurement Method
Clean the Pycnometer with purified water and dry it thoroughly.
Place it vertically in a beaker of a constant-temperature water bath maintained at 30 °C, and ensure that the capillary tube and buffer bulb are fully submerged in the thermostated water.
For each solution sample, draw 10 ml into the storage bulb of the viscometer.
Using a bulb syringe, slowly press the solution into the buffer bulb above the capillary tube, ensuring the liquid level is slightly above the upper scale line.
Remove the bulb. Allow the solution in the buffer bulb to descend slowly under gravity.
Start timing when the meniscus aligns with the upper scale line. Stop timing when the meniscus aligns with the lower scale line.
The longer the solution takes to pass through the capillary, the greater the viscosity (μ). The expression for μ is as follows:
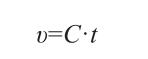
In the equation: C is the viscometer constant, mm²/s²; t is the time, in seconds, taken for the entire solution in the viscometer’s measuring sphere to pass through the capillary tube.
2. Determination of Etching Rate
Measure 1,000 mL of etching solution sample into a beaker.
Place it on a constant-temperature magnetic stirrer, heat to 50°C, and maintain the temperature.
Set the stirring speed to 200 rpm. Before etching, the mass of the copper-clad laminate is m₁,g.
Quickly immerse the weighed copper-clad laminate vertically into the etching solution and start timing.
After 60 seconds, rapidly remove it and immediately rinse thoroughly.
After drying, weigh the copper-clad laminate, recording its mass as m2 ,g. The etching rate is expressed as follows:
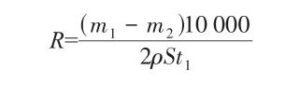
In the formula: P is the density of metallic copper, g/cm³; S is the area of the copper-clad laminate, cm²; t₁ is the etching time, min; R is the etching rate, μm/min.
Results and Analysis
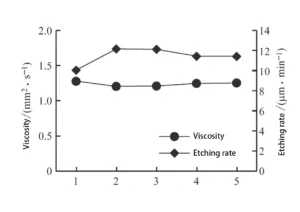
Effect of Different Chloride Salts on Etching Solution Viscosity and Etching Rate
The researchers prepared several etching solution samples using sodium chloride, potassium chloride, and ammonium chloride as chlorides, combined with copper chloride and hydrochloric acid.
The copper ion concentration, acidity, and chloride ion concentration were kept constant across all solutions.
The researchers then measured the etching rates and viscosity values for each sample (see Table 1).
| Etching Solution Formula No. | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Copper(II) chloride, CuCl₂ / (mol·L⁻¹) | 2 | 2 | 2 | 2 | 2 |
| Hydrochloric acid, HCl / (mol·L⁻¹) | 2 | 2 | 2 | 2 | 2 |
| Sodium chloride, NaCl / (mol·L⁻¹) | 1 | — | — | 0.5 | 0.5 |
| Potassium chloride, KCl / (mol·L⁻¹) | — | — | — | 0.5 | — |
| Ammonium chloride, NH₄Cl / (mol·L⁻¹) | — | — | 1 | — | 0.5 |
| Etching rate / (μm·min⁻¹) | 10.05 | 12.15 | 12.11 | 11.43 | 11.41 |
| Viscosity / (mm²·s⁻¹) | 1.278 | 1.204 | 1.210 | 1.247 | 1.253 |
Figure 1 compares the etching rates and viscosities of etching solutions with different chloride salt compositions.
As shown in Figure 1, lower etching solution viscosity correlates with higher etching rates.
Replacing sodium chloride in the etching solution with equivalent molar quantities of potassium chloride or ammonium chloride effectively reduces viscosity and enhances etching rates.
Potassium chloride or ammonium chloride can be used in combination with sodium chloride.
Under identical stirring intensities, lower solution viscosity facilitates faster and more uniform mass transfer within the solution.
Reduced viscosity promotes contact between Cu²⁺ and Cl⁻ ions in the etching solution and the copper metal surface, thereby enhancing the etching rate.
Comparison of Viscosity Among Different Chloride Solutions
Solutions of various chlorides and mixed solutions were prepared, with chloride ion concentration fixed at 3 mol/L. Viscosity values were measured for each (see Table 2).
| Solution No. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Copper(II) chloride (1/2), CuCl₂ / (mol·L⁻¹) | — | 3 | — | — | — | — | — | — |
| Hydrochloric acid, HCl / (mol·L⁻¹) | — | — | 3 | — | — | — | — | — |
| Sodium chloride, NaCl / (mol·L⁻¹) | — | — | — | 3 | — | — | 1.5 | 1.5 |
| Potassium chloride, KCl / (mol·L⁻¹) | — | — | — | — | 3 | — | 1.5 | — |
| Ammonium chloride, NH₄Cl / (mol·L⁻¹) | — | — | — | — | — | 3 | — | 1.5 |
| Viscosity / (mm²·s⁻¹) | 0.802 | 1.048 | 0.933 | 0.997 | 0.758 | 0.775 | 0.855 | 0.868 |
The viscosity values of 1/2 mol/L CuCl₂, HCl, NaCl, KCl, NH₄Cl solutions and pure water, arranged from highest to lowest, are: CuCl₂, NaCl, HCl, water, NH₄Cl, KCl.
Hydration and hydrolysis interactions between ions and water molecules alter the original arrangement of water molecules to some extent, thereby inducing viscosity changes.
Typically, ions with weak electric field strengths and large radii—such as K⁺, Rb⁺, NH₄⁺, Cl⁻, I⁻, and NO₃⁻—exhibit weak hydration, resulting in low solution viscosity.
Conversely, ions like Cu²⁺, Fe³⁺, Li⁺, Na⁺, and F⁻ undergo strong hydration, leading to high solution viscosity.
Moreover, the higher-valent ions among these can undergo hydrolysis reactions, further increasing solution viscosity.
An equimolar potassium chloride solution exhibits higher conductivity than a sodium chloride solution at the same temperature.
This occurs because K⁺ ions are less strongly hydrated than Na⁺ ions, resulting in a thinner hydration layer and lower electromigration resistance.
Experiments reveal that copper chloride dihydrate dissolves with slight endothermic heat release, while sodium chloride exhibits negligible heat absorption or release during dissolution.
Conversely, potassium chloride and ammonium chloride dissolve with intense endothermic heat absorption.
This phenomenon aligns with the pattern of hydration strength among corresponding ions.
Generally, stronger hydration tends to favor exothermic reactions during dissolution.
Effect of Concentration on Solution Viscosity
A series of solutions with varying concentrations of 1/2CuCl₂, NaCl, and KCl were prepared.
The researchers measured their viscosity values to investigate how concentration changes affect the viscosity of different chloride solutions (see Table 3).
| Concentration / (mol·L⁻¹) | 0 | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| 1/2 CuCl₂ solution viscosity / (mm²·s⁻¹) | 0.802 | 0.891 | 0.961 | 1.048 | 1.145 |
| NaCl solution viscosity / (mm²·s⁻¹) | 0.802 | 0.848 | 0.910 | 0.997 | 1.100 |
| KCl solution viscosity / (mm²·s⁻¹) | 0.802 | 0.781 | 0.768 | 0.758 | 0.762 |
For high-viscosity solutions such as copper chloride and sodium chloride, viscosity increases with rising concentration.
For low-viscosity solutions like potassium chloride, viscosity decreases gradually with increasing concentration at low concentrations, then stabilizes as concentration continues to rise.
Therefore, adding potassium chloride or ammonium chloride to the etching solution will not cause significant fluctuations in viscosity.
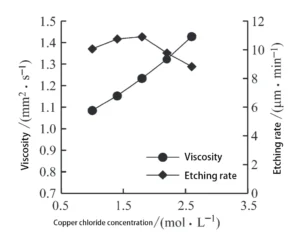
Effect of Copper Ion Concentration on Viscosity and Etching Rate
Based on the above experimental results, it is evident that among the etching solution components, copper ion concentration exerts the most significant influence on solution viscosity.
A series of etching solution samples were prepared with fixed concentrations of hydrochloric acid (2 mol/L) and sodium chloride (1 mol/L), while varying the copper chloride concentration.
The researchers measured the etching rate and viscosity values for each sample (see Table 4).
| Parameter | Unit | Solution 1 | Solution 2 | Solution 3 | Solution 4 | Solution 5 |
|---|---|---|---|---|---|---|
| Copper(II) chloride (CuCl₂) | mol·L⁻¹ | 1.0 | 1.4 | 1.8 | 2.2 | 2.6 |
| Hydrochloric acid (HCl) | mol·L⁻¹ | 2 | 2 | 2 | 2 | 2 |
| Sodium chloride (NaCl) | mol·L⁻¹ | 1 | 1 | 1 | 1 | 1 |
| Etching rate | μm·min⁻¹ | 10.06 | 10.75 | 10.91 | 9.78 | 8.83 |
| Viscosity | mm²·s⁻¹ | 1.085 | 1.153 | 1.234 | 1.323 | 1.428 |
› Relationship Between Copper Chloride Concentration, Viscosity, and Etching Rate
Figure 2 shows the variation in etching solution viscosity and etching rate at different copper chloride concentrations.
As shown in Figure 2, the viscosity of the etching solution increases continuously with rising copper chloride concentration.
Meanwhile, the etching rate initially increases but then rapidly decreases after reaching a certain level.
Cu²⁺ ions possess high charge density and undergo both hydration and hydrolysis in aqueous solutions, exhibiting strong interactions with water molecules.
Consequently, copper chloride concentration exerts the most pronounced effect on viscosity.
As one of the reactants, variations in copper ion concentration exert a complex influence on the etching reaction process.
At low copper ion concentrations, increasing the reactant copper ion concentration accelerates the etching rate, making reactant concentration the primary factor influencing reaction rate.
Elevated copper ion concentration increases the etching solution’s viscosity, thereby inhibiting mass transfer during the etching reaction.
Under these conditions, viscosity dominates the etching rate, causing it to decrease.
› Chloride Ion Depletion and Complex Formation
During etching, copper ions and chloride ions combine as complexes. Higher copper ion concentrations reduce the free chloride ion concentration in the etching solution.
However, the monovalent copper produced by the etching reaction can only remain stable by forming complexes with chloride ions.
When free chloride ion concentration becomes excessively low, insoluble copper chloride precipitates may even form on the copper surface, further inhibiting the etching reaction.
At the interface between the etching solution and the copper metal, the concentration of monovalent copper rapidly increases, forming low-etching-capacity, high-viscosity etching waste liquid.
This waste liquid obstructs contact between the etching solution and the copper metal, thereby reducing the etching rate.
This phenomenon manifests particularly severely on the top surface of the board during the etching production process, known as the “pooling effect.”
High-copper etching solutions exhibit strong adhesion to the copper surface, making the pooling effect even more pronounced.
Effect of Temperature on Etching Solution Viscosity
Two etching solution samples were prepared: one with a copper chloride concentration of 2 mol/L and hydrochloric acid concentration of 2 mol/L, and the other with sodium chloride and potassium chloride each at 1 mol/L.
These were designated as the NaCl-type etching solution and KCl-type etching solution, respectively.
The viscosity of both etching solutions was measured under different temperature conditions (see Table 5).
| Parameter | Unit | 20°C | 30°C | 40°C | 50°C |
|---|---|---|---|---|---|
| Viscosity of NaCl-type etching solution | mm²·s⁻¹ | 1.521 | 1.277 | 1.037 | 0.870 |
| Viscosity of KCl-type etching solution | mm²·s⁻¹ | 1.458 | 1.210 | 50.984 | 0.830 |
The viscosity values of both etching solutions decrease with increasing temperature, and the KCl-based etching solution consistently exhibits lower viscosity.
On one hand, elevated temperatures intensify the thermal motion of solute ions and water molecules.
This inhibits ion hydration and weakens hydrogen bonding forces between water molecules, thereby reducing solution viscosity.
On the other hand, increased temperature enhances the chemical reactivity of the reactants.
Consequently, temperature elevation promotes faster reaction rates.
Effect of Polyethylene Glycol (PEG) on Etching Solution Viscosity
An etching solution containing 2 mol/L copper chloride, 2 mol/L hydrochloric acid, and 1 mol/L sodium chloride was divided into equal portions.
Different amounts of polyethylene glycol 6000 (PEG 6000) were added to each portion, and their viscosities were subsequently measured (see Table 6).
As shown in Table 6, the viscosity of the etching solution increases with rising PEG content.
| PEG 6000 Concentration (×10⁶) | 0 | 50 | 100 | 500 | 1,000 |
|---|---|---|---|---|---|
| Viscosity (mm²·s⁻¹) | 1.278 | 1.278 | 1.282 | 1.291 | 1.314 |
During the electrolytic regeneration and reuse of etching waste liquid, a certain amount of PEG (molecular weight above 4,000) is added to improve the quality of electrolytic copper.
With prolonged recycling, PEG gradually accumulates in the etching solution.
PEG itself possesses a macromolecular structure, with numerous oxygen atoms on its molecular chains forming hydrogen bonds with water molecules.
This leads to increased viscosity of the etching solution, consequently reducing etching rates.
On one hand, when electrolyzed recycled solution is reused in the etching production line, it typically results in a 5% to 10% reduction in the original etching rate.
On the other hand, during the etching production process, organic impurities are formed by the dissolution of ink and other substances on the PCB surface.
These impurities also contribute to the increased viscosity of the etching solution.
Conclusion
Under constant reactant concentrations and reaction conditions in the etching solution, lower solution viscosity correlates with faster etching rates.
Measures such as substituting potassium chloride or ammonium chloride for sodium chloride can reduce solution viscosity.
Maintaining optimal copper chloride concentrations also helps.
In addition, elevating etching temperatures and removing organic impurities further reduce viscosity and enhance etching rates.
Complex microscopic interactions within the solution—such as hydration, hydrolysis, and hydrogen bonding—all influence the efficiency of ion migration and mass transfer, thereby affecting the etching rate.
Viscosity serves as a macroscopic indicator of microscopic mass transfer resistance and is readily quantifiable.
Consequently, studying etchant viscosity offers strong practicality and applicability in formulating etchant recipes and developing etching additives.


