Printed circuit boards (PCBs) serve as the medium for electrical interconnection among numerous electronic components, earning them the title of “mother of electronic devices.”
Since Albert Hanson proposed the concept of forming circuits by placing conductive foil on insulating substrates in 1903, the foundation of printed circuit technology was established.
In 1936, Paul Eisner further advanced this idea and formally developed the manufacturing technology for printed circuit boards.
Since then, PCBs have undergone decades of continuous and significant development.
They have evolved from simple single-sided boards to complex multilayer high-density interconnect boards, greatly advancing the progress of the electronics industry.
Today, diverse PCB products are extensively utilized in computing, communications, aerospace, medical technology, and high-end digital sectors, becoming indispensable components in all electronic devices.
Electroless Copper Plating in PCBs
Electroless copper plating, commonly known as immersion copper, is a process in which manufacturers chemically deposit a 2–3 μm thin copper layer onto the insulated hole walls of printed circuit boards.
This process enables electrical interconnection between different PCB layers or between copper foils.
The deposition quality of this thin copper layer directly determines the PCB’s electrical connection performance.
During PCB electroless copper plating, manufacturers may encounter several defects.
These include copper discontinuity on hole walls, rough copper surfaces, uneven copper thickness, cracks, and separation between the copper layer and the hole wall.
Such defects usually arise from insufficient production equipment capability or outdated plating technology.
PCBs must withstand high-temperature reflow soldering during electronic component assembly and may endure repeated thermal shock during normal operation.
High-frequency circuit boards used in satellite communications or data servers impose stringent requirements on the deposited copper layer’s uniformity, surface roughness, adhesion, and thermal shock resistance.
Amid ongoing advancements in PCB manufacturing capabilities, the industry remains committed to developing cost-effective chemical copper plating processes that deliver reliable performance.
Impact of Magnetic Fields on Chemical Deposition
As a vital branch of modern physics, magnetism research indicates that all matter possesses specific magnetic properties.
These properties primarily consist of orbital magnetic moments generated by electrons orbiting atomic nuclei and spin magnetic moments arising from the intrinsic rotation of electrons.
The influence of magnetic fields on chemical reactions has also attracted extensive scholarly investigation.
Their observations revealed that the silver dendrite morphology transformed from the typical diffusion-limited aggregation (DLA) pattern into a dense vortex structure.
The cobalt content in the film progressively increased, while the tungsten and phosphorus contents decreased.
At higher magnetic field strengths, deposition accelerated significantly.
The magnetic field also promoted cobalt atom growth along the (002) crystal orientation.
As a result, the film formed a hexagonal cobalt (c-Co) structure.
Results indicated that under strong magnetic fields, nickel nanowires exhibited smooth surfaces and smaller diameters, whereas under weak magnetic fields, the nanowires displayed rough surfaces, larger diameters, and sawtooth structures.
Lower magnetic field strengths correlated with larger nanowire diameters and more irregular morphologies.
Han Jinru et al. observed that applying an external magnetic field during the thermite reduction process for producing CuC(r 50) alloy enabled the CuCr(50) alloy melt to overcome the tendency for high-Cr-content CuC(r 50) alloys to stratify.
This resolved segregation issues in CuC(r 50) alloys, achieving homogenization of the alloy.
Research Gap and Study Focus
Kołodziejczyk et al. summarized extensive research on magnetic field effects in metal deposition, reviewing the diverse impacts of constant magnetic fields on the deposition processes of metals, including copper, zinc, nickel, iron, and cobalt.
These examples demonstrate the regulatory role of high magnetic field strength on deposition rate, morphology, and crystal growth orientation in chemical deposition processes.
However, few reports exist on chemical copper deposition behavior under low magnetic field conditions (<0.1 T).
This study investigates the effect of low-gradient magnetic fields on copper plating performance for PCBs using widely applied rigid PCBs as the research subject.
We conduct rate measurements, morphology characterization, and electrochemical testing of chemical copper plating processes under different magnetic field conditions.
Through these investigations, we aim to improve the economic reliability of copper plating layers. We also seek to advance PCB copper plating technology.
Experiment
Electroless Copper Plating Process
The experiment employed a gantry-type copper plating production method. The process flow was as follows:
Hole cleaning → Water rinse → Micro-etching → Water rinse → Pre-soaking → Activation → Water rinse → Acceleration → Water rinse → Electroless copper plating.
Key process parameters and reagents used are listed in Table 1.
All chemical reagents used in the experiment were analytical grade (AR grade) products manufactured by Xilong Science Co., Ltd.
Other commercial reagents were supplied by Xinfeng Zhengtianwei Technology Co., Ltd.
We used a 25 mm × 25 mm × 1 mm Flame Retardant 4 (FR-4) board as the substrate for chemical copper plating.
This material primarily consists of glass fiber and epoxy resin and is commonly used in PCB production.
We used a 30 mm × 20 mm × 10 mm neodymium iron boron magnet as the external magnetic field source.
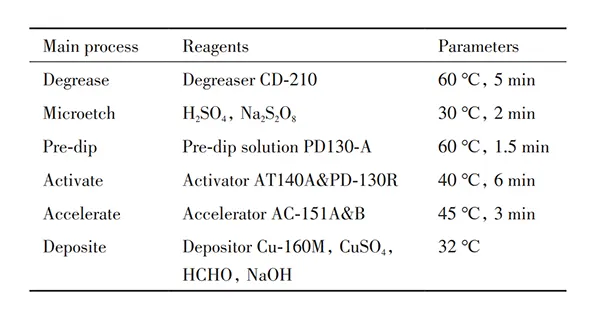
Experimental Method
A beaker was used as the chemical copper plating tank to prepare the required plating solution, which was placed in a constant-temperature water bath (Jiangsu Zhongda Instrument Co., Ltd.).
Copper plating operations were strictly conducted according to the process parameters for each step.
The copper plating setup is illustrated in Figure 1.
A neodymium iron boron magnet was fixed externally to the plating tank as an external magnetic field source.
The internal magnetic field strength was adjusted by altering the tank size (volumes of 100 and 25 ml) to change the distance from the permanent magnet.
Magnetic field strength is expressed as magnetic flux density.
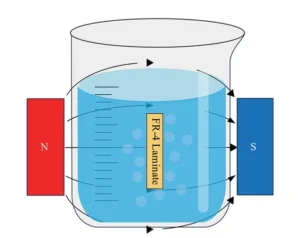
A Gauss meter (TD8620, Changsha Tianheng Measurement & Control Technology Co., Ltd.) was used to measure field strength at different positions within the tank.
The measured field distribution results are shown in Figure 2.
Substrate Preparation and Deposition Rate
The internal magnetic field is not symmetrically distributed; instead, the lower section exhibits slightly higher magnetic field strength than the upper section.
During electroless copper plating, fixtures were used to secure the substrates at the center of the tank.
The center field strength was 30 mT for the 100 ml tank and 60 mT for the 25 ml tank.
Before electroless copper plating, each substrate was weighed using a precision analytical balance (FA-2004, Shanghai Shangping Instrument Co., Ltd.).
After copper plating in different magnetic field environments, substrates were rinsed, dried, and reweighed.
The copper deposition rate (v) was calculated according to Equation (1):

In the equation, Δm represents the mass difference of the substrate before and after copper plating, in mg.
A denotes the actual plated area of the substrate, in dm²; and t indicates the copper plating time of the substrate, in min.
Analysis of Copper Layer
The microstructure of the copper layer on the plated substrate under different magnetic field conditions was observed using a field emission scanning electron microscope (SEM, JSM-6510A, JEOL Ltd.).
Electrochemical testing was conducted using an electrochemical workstation (Shanghai Chenhua CHI660E) in a conventional three-electrode system.
Cyclic voltammetry was employed to investigate the effect of applied magnetic fields on the copper ion deposition process in solution.
Additionally, AC impedance spectroscopy was utilized to analyze the corrosion resistance of the copper layer on the plated substrates under different magnetic field conditions.
Results and Discussion
Effect of Magnetic Field on Copper Deposition Rate
We conducted three copper plating experiments under identical process conditions in different magnetic field environments.
Deposition rate measurements were conducted using the previously described weighing method.
We then calculated the average values of the three trials.
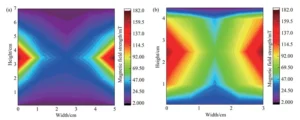
Finally, we plotted these averages to illustrate the relationship between copper deposition rate and plating time, as shown in Figure 3.
Deposition Rate Trends and Magnetic Field Effect
The results indicate that regardless of magnetic field application, the copper deposition rate on the substrate exhibits a trend of initially increasing and then decreasing as plating time extends.
The deposition rate peaks between 2 and 3 minutes after the reaction begins, then gradually decreases over time until reaching a stable range.
When magnetic fields of varying intensities were applied externally to the plating tank, the overall deposition rate significantly increased compared to the condition without an external magnetic field.
Considering the stable rate interval from 3 to 7 minutes after copper plating, the rate increased from approximately 8 mg·dm⁻²·min⁻¹ without a magnetic field to 10 mg·dm⁻²·min⁻¹ with one, representing a 25% improvement.
Impact of Tank Size
Figure 3 also reveals substantial differences in copper deposition rates during the pre-stabilization phase across tanks of varying capacities.
During this phase, the copper deposition rate on substrates in smaller tanks is significantly higher than that in larger tanks.
This is because the deposition behavior of copper ions in the solution is primarily governed by their own concentration and catalytic reactivity.
When substrates are immersed in tanks of different capacities, the effective concentration of active copper ions surrounding them during the early deposition stage differs, leading to noticeable variations in deposition rates.
Effect of Magnetic Fields on Copper Layer Surface Morphology
Figures 4 and 5 show the differences in appearance between the copper-plated layers on substrates before and after applying magnetic fields of 30 mT and 60 mT, respectively.
It can be observed that during copper plating, the substrate’s copper layer gradually changes from a dull appearance to a bright, powdery finish.
The application of a magnetic field significantly accelerates this process.
When we apply the magnetic field, the copper layer appears brighter and more vivid than under the non-magnetic field condition.
This difference is particularly pronounced during the first three minutes of plating.
In the absence of a magnetic field, the surface of the plated substrate appears slightly rough, and minor plating defects can be observed when the plating time is less than 2 minutes.
Increasing the magnetic field strength from 30 mT to 60 mT further accentuates these phenomena, indicating that heightened magnetic field intensity promotes the formation of a more vivid and lustrous surface copper layer.
To further determine the magnetic field’s impact on copper layer surface morphology, SEM was employed to examine the microscopic structure of the copper layer.
SEM Analysis of Copper Layer Morphology
Figure 6 displays SEM images of the surface copper layer after 300 and 420 seconds of electroless copper plating under different magnetic field conditions.
When the copper layer on the substrate surface is thin or uneven, the substrate morphology becomes visible through the surface copper layer in the SEM images.
It can be observed that, in the absence of a magnetic field, the deposited copper layer is sparsely distributed and loose, with the underlying substrate morphology visible in some areas (Figure 6(a, b, e, f)).
After applying external magnetic fields of 30 mT (Fig. 6(c, d)) and 60 mT (Fig. 6(g, h)), copper ions deposited uniformly on the substrate surface, with significantly enhanced density and uniformity.
.webp)
.webp)

Effect of Magnetic Fields on Copper Ion Deposition Process
Copper sulfate solutions at different concentrations (referencing the copper plating process for printed circuit boards, where the optimal copper ion concentration is 2–5 g·L^(−1)) were prepared.
A glassy carbon electrode served as the working electrode, a platinum plate electrode as the counter electrode, and a Hg/Hg₂SO₄ electrode as the reference electrode.
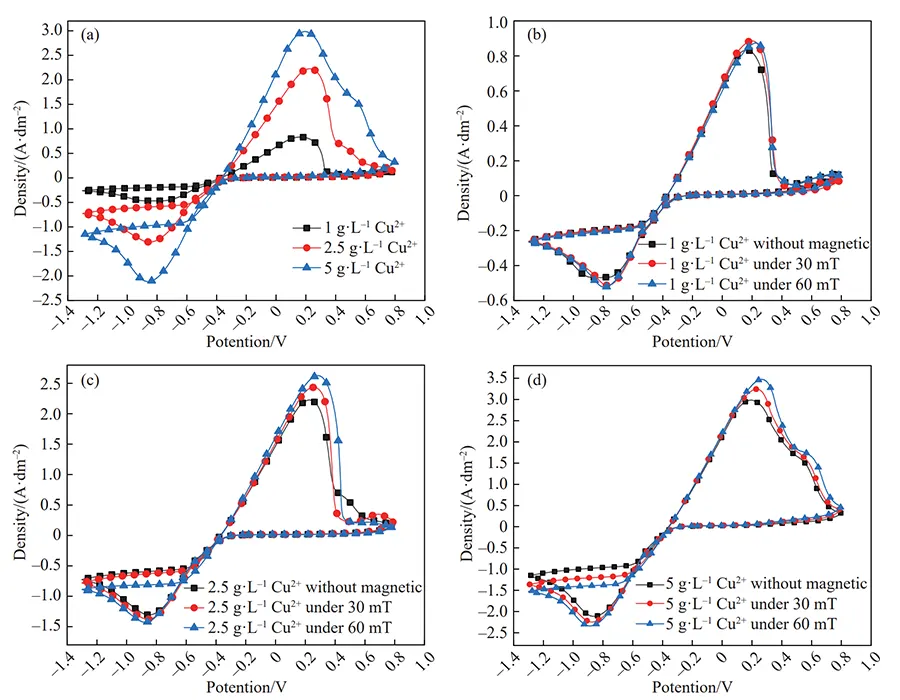
Cyclic voltammetry (CV) was employed to investigate changes in the copper ion deposition process under an applied magnetic field. The CV curves are shown in Figure 7.
Effect of Copper Ion Concentration on Deposition Behavior
It is evident that in the absence of a magnetic field, both the reduction and oxidation peak areas significantly increase with rising copper ion concentration.
The reduction peak current density rises from 0.47 A·dm⁻² to 2.11 A·dm⁻², indicating that copper deposition remains primarily concentration-controlled.
As shown in Figure 7(b), the CV curve of a 1 g·L−1 copper ion solution under an applied magnetic field nearly overlaps with that without a magnetic field.
This indicates that the effect of the applied magnetic field can be neglected at low copper ion concentrations.
Influence of Magnetic Field on Copper Ion Reduction
As the copper ion concentration was successively increased to 2.5 and 5 g·L−1 (Fig. 7(c, d)), the reduction peak potentials of copper ions under an applied magnetic field exhibited varying degrees of negative shift.
The negative shift of the reduction peak potential became more pronounced with increasing copper ion concentration and magnetic field strength.
Under a 60 mT magnetic field, the reduction peak potential of the 5 g·L−1 copper ion solution shifted negatively from −0.85 V to −0.89 V, while the peak current density increased from 2.11 A·dm⁻² to 2.34 A·dm⁻².
That is, the applied magnetic field caused a negative shift in the copper ion reduction peak potential and increased the cathode limiting current density.
This indicates that the magnetic field enhances cathode polarization, facilitating faster nucleation rates and slower crystal growth during copper ion deposition at the cathode.
Consequently, the deposited copper lattice becomes denser and smoother. while the increased limiting current density also permits a faster deposition rate of copper ions.
Differences in Copper Deposition Layer Corrosion Under Varied Magnetic Field Conditions
We used copper-plated substrates exposed to different magnetic field conditions as the working electrodes.
The saturated calomel electrode was used as the reference electrode.
We employed a dilute hydrochloric acid solution with a pH of 4 as the corrosion medium.
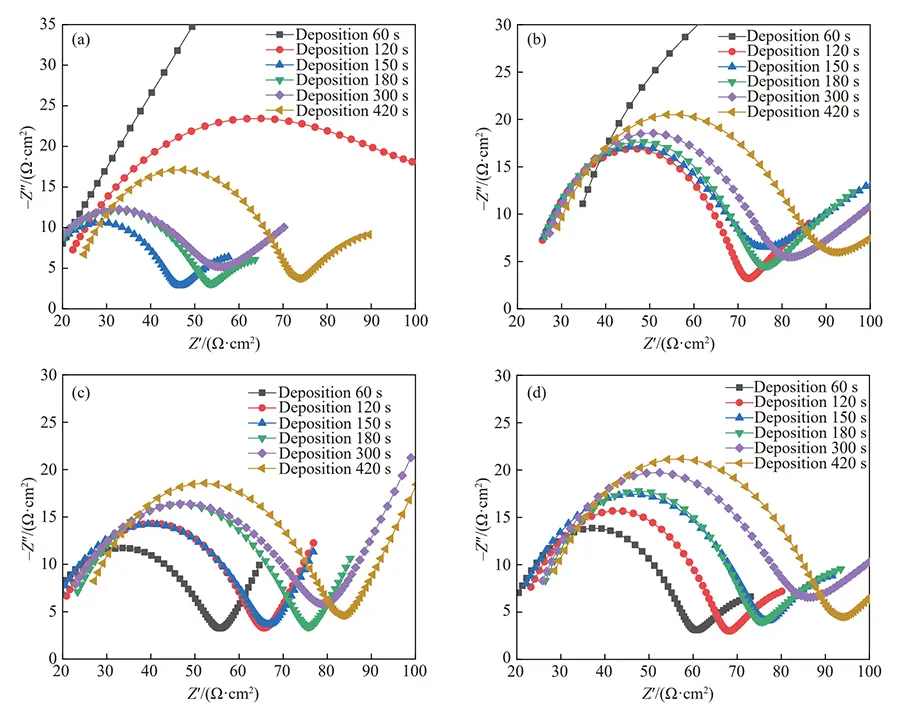
Under these conditions, we conducted AC impedance spectroscopy (EIS) tests. The results are shown in Figure 8.
Interpretation of EIS Spectra
The radius of the capacitive arc in the EIS spectrum reflects the corrosion resistance of the plating layer.
A larger radius indicates higher charge transfer resistance and better corrosion resistance.
However, the copper layers deposited for 60 and 120 seconds in Figure 8(a) and the 60-second layer in Figure 8(b) did not exhibit distinct capacitive arcs.
This indicates that these copper layers possessed poor corrosion resistance, having been corroded during testing to expose the underlying substrate, thus preventing completion of the full EIS test.
Effect of Plating Time and Tank Size
By comparing the radii of the capacitive arc in normal spectra, it is evident that longer copper plating times result in thicker surface copper layers, more stable deposition, and stronger corrosion resistance.
Furthermore, copper layers deposited in small tanks exhibit superior corrosion resistance compared to those in large tanks.
Influence of Magnetic Field on Corrosion Resistance
After we applied an external magnetic field to the copper plating bath (Fig. 8(c, d)), the copper layers on substrates with shorter plating times were also able to complete the entire AC impedance test.
These samples exhibited a typical impedance spectrum shape.
Compared with the condition without a magnetic field, the capacitive arc of each deposited copper layer increased to varying degrees under the magnetic field.
When the magnetic field strength increased from 30 mT to 60 mT, the radius of the capacitive arc also increased to varying degrees.
These results indicate that externally applied magnetic fields of different strengths can enhance the corrosion resistance of the copper layer deposited on the substrate.
Analysis of the Mechanism by Which Magnetic Fields Affect Electrolytic Copper Plating Performance
1. Mechanism of Electrolytic Copper Plating Process
The chemical copper plating process can be divided into two steps: substrate pretreatment and chemical copper plating.
The purpose of pretreatment is to remove contaminants such as oil, dust, and oxides from the board surface while adjusting the polarity of the insulating substrate (from negative to positive charge).
It also involves mild etching of the insulating wall to increase its roughness, thereby enhancing the adhesion and adsorption area for subsequent copper deposition.
This creates pore wall activation centers conducive to electroless copper plating.
Electroless copper plating is a typical catalytic redox reaction, typically catalyzed by metallic palladium but also capable of autocatalytic reactions involving metallic copper, as illustrated in Figure 9.
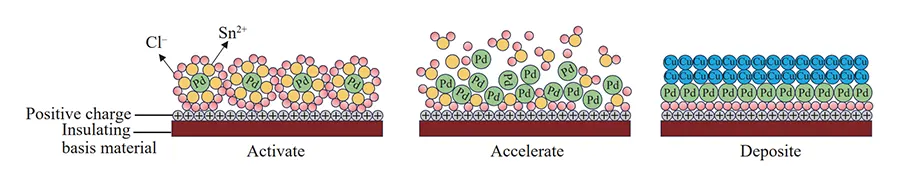
The pretreated insulating walls carry a positive charge, enabling adsorption of colloidal palladium particles that are negatively charged as a whole;
The periphery of the colloidal palladium core is enveloped by Sn²⁺ and excess Cl⁻ ions.
The outer layer of negative charges repels each other, preventing colloidal particle aggregation and sedimentation.
Upon immersion in the acceleration solution, the outer ions of the colloidal palladium dissociate, exposing the highly catalytic inner atomic palladium.
Copper ions in the copper plating bath are then reduced and deposited onto the exposed colloidal palladium core surface through the synergistic action of formaldehyde, sodium hydroxide, and complexing agents.
The main reaction equation for electroless copper plating is:
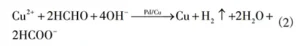
As seen from equation (2), the reduced copper atoms themselves also exhibit catalytic activity, allowing the copper deposition reaction to continue on the surfaces of newly formed copper nuclei.
However, palladium possesses significantly higher catalytic activity than copper.
Consequently, the copper deposition rate described in Section 2.1 will peak within 2–3 minutes before gradually decreasing to a steady-state value.
2. A Brief Analysis of the Mechanism of Magnetic Fields on Copper Deposition
Researchers have long explored how magnetic fields influence the physicochemical properties of materials.
They have successfully demonstrated that externally applied magnetic fields can directionally regulate chemical reactions.
Based on these findings, researchers have proposed several mechanism models.
These include the Lorentz force theory, the free radical pair theory, and the magnetic bond theory.
However, due to the numerous influencing factors and complex mechanical properties of magnetic fields, existing hypotheses cannot fully elucidate the mechanism of magnetic field effects on chemical reactions.
Analysis suggests that magnetic fields may influence copper deposition performance by exerting Lorentz forces and magnetic gradient forces on charged ions, thereby inducing magnetohydrodynamic (MHD) effects at the chemical reaction interface.
This mechanism is illustrated schematically in Figure 10.
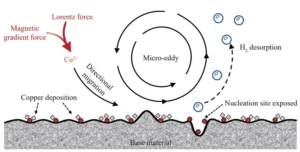
When paramagnetic Cu²⁺ moves in a non-uniform magnetic field, it experiences both the Lorentz force and the magnetic gradient force.
The combined effect of these forces drives Cu²⁺ to migrate directionally from the solution toward the reaction interface within the magnetic field.
This induces the formation of micro-eddy currents (micro-MHD effect).
This micro-MHD effect enhances convection within the plating solution, generating micro-stirring action that reduces diffusion layer thickness, enhances ion transport, and accelerates local deposition rates.
Furthermore, during Cu²⁺ deposition, ions first adsorb onto active sites at the reaction interface to form crystal nuclei, which then gradually grow into crystals.
During this process, the nucleation rate exceeds the crystal growth rate, favoring the formation of a denser deposition layer.
Micro-vortices generated at the reaction interface accelerate the detachment of hydrogen gas bubbles produced during copper deposition, reducing hindrance to Cu²⁺ transport and exposing more nucleation sites;
The increased number of nucleation sites leads to finer grains, enhancing the uniformity and density of the deposited layer.
Conclusion
1) Applying a low external magnetic field during the electroless copper plating process on printed circuit boards significantly enhances the copper deposition rate.
This accelerates the transformation of the plated copper surface from a dull appearance to a bright, powdery finish, resulting in a more vivid and lustrous plated copper layer.
2) Under non-uniform magnetic fields, copper ions undergo deposition influenced by both Lorentz forces and magnetic gradient forces.
This induces micro-MHD effects at the reaction interface, accelerating nucleation while reducing crystal growth rates.
Consequently, the deposited layer exhibits improved uniformity and density, yielding a copper plating layer with enhanced corrosion resistance.


